The multiple award-winning treatment developed by the Lausanne University Hospital and University of Geneva will launch clinical trials in 2016. The drug offers such promise that a British pharmaceuticals firm has already picked up the licence for its manufacture.
PACTT website: http://www.pactt.ch/
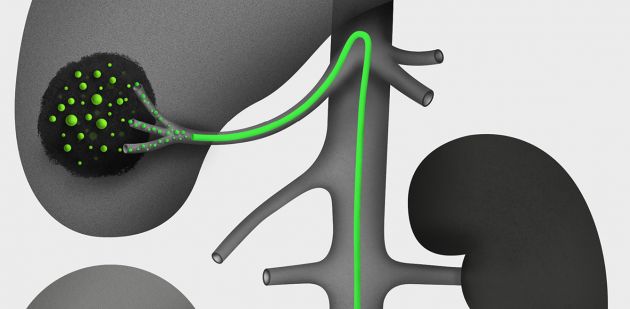
If we had to perform a grim ranking of cancer, liver cancer would score top marks. In sixth place in terms of prevalence, the disease comes in as third deadliest. The new therapy developed in partnership between the Faculty of Pharmaceutical Sciences at the University of Geneva and the Service of Interventional Radiology at the Lausanne University Hospital (CHUV) could affect a large number of patients.
The idea is simple, starving liver tumours to death. This forced regimen works on two fronts. First, embolisation spheres stop blood flow to diseased cells by blocking the capillary arteries in the liver. Then the beads release an anti-angiogenic agent to prevent the development of new blood vessels. This method is both more targeted and more effective than current forms of chemotherapy. “All the preclinical toxicity and efficacy tests were conducted successfully. Now we’re moving into the clinical phase,” says Professor Alban Denys, chief of the Department of Interventional Radiology at CHUV.
As proof of the new therapy’s potential even at this early development stage, the patent filed with the support of PACTT, the technology transfer office at CHUV, has been licensed to the British pharmaceutical group BTG. Professor Denys and his research team (Pierre Bize from CHUV and Katrin Fuchs, Olivier Jordan and Professor Gerrit Borchard from the University of Geneva) have received the CIRSE Award of Excellence and Innovation in Interventional Radiology and the 2014 JVIR Editor’s award for Outstanding Laboratory Investigation for their achievements.
“This kind of recognition is rare in a researcher’s life,” Alban Denys says. Years and years of research separate the idea from the new drug. In between, the patent, administrative procedures and partnerships within the pharmaceutical industry are the compulsory but essential steps towards pushing the bounds of research further.”